Laboratory of Human Molecular Genetics Research Interests
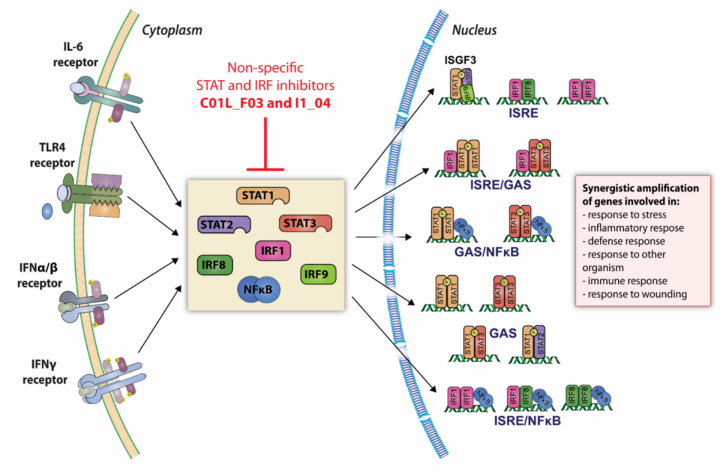
Key factors contributing to anti-pathogen responses and inflammation include type I and type II interferons (IFNs) and activators of Toll-like receptor 4 (TLR4). Together, they trigger activation of members of the Signal Transducer and Activator of Transcription (STAT) and Interferon Regulatory Factor (IRF) family. In particular STAT1, 2 and 3 and IRF1, 8 and 9 have recently been recognized as prominent modulators of these responses.
(1) TheLaboratory of Human Molecular Genetics aims at genome-wide and mechanistic understanding of the STAT- and IRF-dependent signal integration between TLR4 and IFNs (Type I and II) in vascular and immune cells in culture, and in experimental and clinical atherosclerosis. Identifying transcriptional mechanisms, pathways and target genes regulated under established conditions of signal integration that correlate with atherosclerosis, should aid the development of novel strategies to predict disease susceptibility, target and monitor therapies, and ultimately develop new approaches to the prevention and treatment of cardiovascular disease (CVD).
Szeląg et al., Oncotarget, 2016 (PubMed)
Chmielewski, Piaszyk-Borychowska et al., IRI, 2016 (PubMed)
Chmielewski et al., PlosOne, 2014 (PubMed)
Sikorski et al., Int J Mol Sci, 2014 (PubMed)
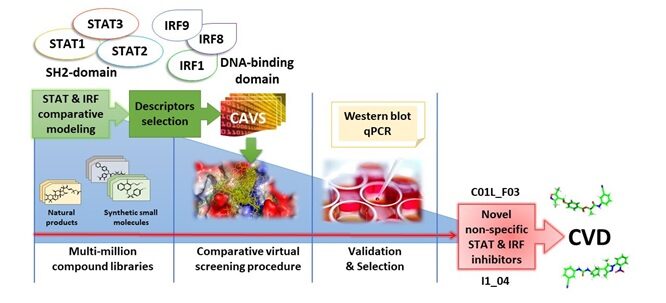
(2) We also develop comparative virtual screening and docking validation tools to identify small inhibitory compounds of STATs and IRFs as a novel therapeutic strategy in the management of CVD.
Plens-Galaska et al ., Scientific Data, 2022 (PubMed)
Plens-Galaska et al., Frontiers in Immunology, 2018 (PubMed)
Szeląg et al., Oncotarget, 2016 (PubMed)
Szeląg et al., PlosOne, 2015 (PubMed)
Szeląg et al., Journal of Computational Science, 2015 (ScienceDirect)
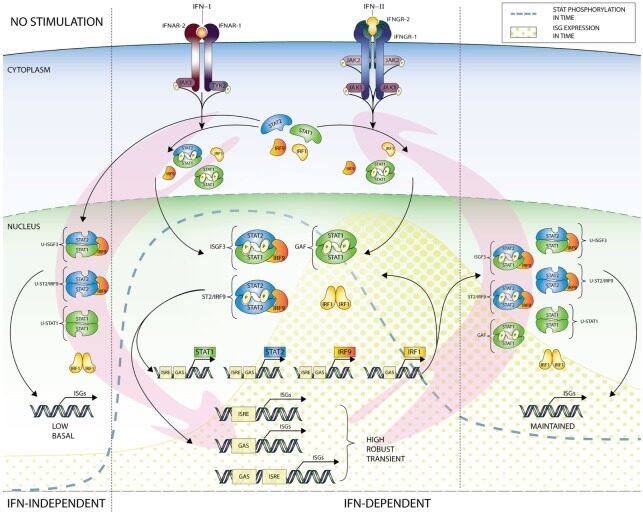
(3) Our research aims at genome-wide understanding of canonical and alternative type I and II IFN-induced responses, to elucidate the full Type I and II IFN-induced transcriptome and chromatin interactions mediated by STAT1, STAT2 and IRF9, and identify known and novel protein coding ISGs and their potential antiviral functions. This may support the identification of novel therapeutic uses for IFNs and novel biomarkers to monitor clinical responsiveness to IFN therapy as well as the development of novel antiviral drugs.
Michalska et al., Frontiers in Immunology, 2018 (PubMed)
Blaszczyk et al., CGFR, 2016 (PubMed)
Blaszczyk et al., Biochem J, 2015 (PubMed)
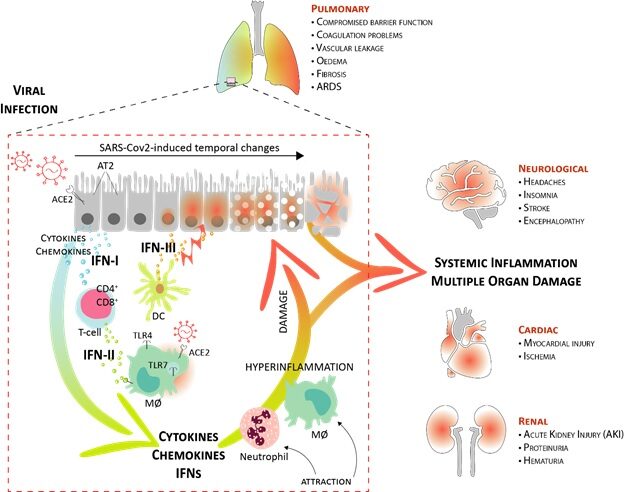
(4) We are studying innate and adaptive immune-associated transcriptional responses of COVID-19 patients in a cell type-specific manner in relation to clinical outcomes. It aims to increase our understanding of the dynamics of the IFN response, dependent on STATs and IRFs, and its possible contribution to the development of severe COVID-19 symptoms in different tissues. Together, this could serve as a molecular framework for developing novel clinical strategies of patient stratification, diagnostics and treatment in the global fight against SARS-CoV-2 infection.
Eskandarian Boroujeni et.al., Frontiers in Immunology, 2022 (PubMed)
Eskandarian Boroujeni et.al., ACS Chemical Neuroscience, 2021 (PubMed)
Laboratory of High Throughput Technologies Research Interests
(1) The main purpose of the LHTT is the management of the sequencing platform (Illumina). The Genome Analysis Laboratory was funded by National Multidisciplinary Laboratory of Functional Nanomaterials NanoFun (project POIG.02.02.00-00-025/09). The platform is available for the investigators of Faculty of Biology, but also all researchers interested in initiation of scientific collaboration.
(2) The main research topic of the Laboratory is genomic characterization of clear cell Renal Cell Carcinoma (ccRCC), with focus on transmembrane proteins and their role in ccRCC etiology. Since molecular markers aiding early diagnosis and monitoring of ccRCC recurrence are virtually non-existent, we concentrate our efforts on identification of mutational and/or expression profiles, that can potentially mirror the stage of the disease.
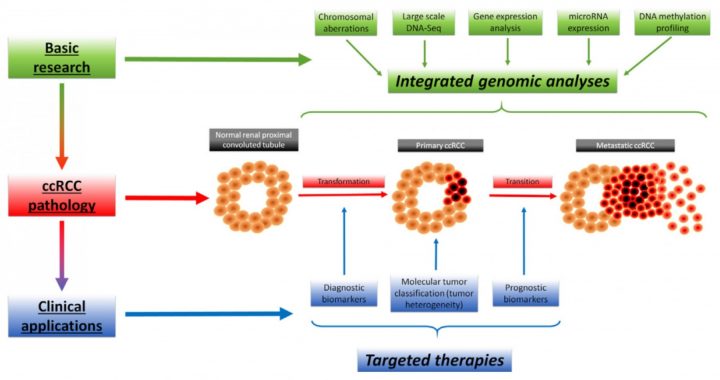
Fig. 1. The concept of integrative genomic approaches in ccRCC management (Rydzanicz M, et al., 2013, PubMed)
